
Switchable adsorption materials for selective separation processes
The patented technology enables a selective adsorption and efficient separation of metal cations and metal-containing anions from complex solutions.
Challenge
While bismuth, silicon, cobalt, aluminum and palladium are listed as critical raw materials in the European Union, elements such as copper, zinc and nickel are of great technological relevance. Separation of metal cations or heavy metal cations is not only relevant for the treatment of drinking water and wastewater. Rather, aqueous solutions also occur after hydrometallurgical digestion of natural ores or slags from recycling processes. In this case, metals may be converted into complex mixtures of various water-soluble anions and cations. Challenges arise when a separation of specific metal cations from aqueous solutions, which contain a large number of cations and anions, is desired. Heavy metals occur in water as cations, more rarely as complex anions. Ion exchangers and membrane absorbers may be used for the filtration of drinking water, while activated carbon is often used for the removal of heavy metals from drinking water, wastewater and process water. Relatively long contact times, which are necessary for diffusion into pores, and large activated carbon quantities required in wastewater treatment plants may be drawbacks from the technological and/or economic point of view.
Our Solution
Scientists from the Clausthal University of Technology have developed low cost, highly stable, switchable materials for a selective adsorption and separation of metal cations and metal-containing anions from complex mixtures and solutions. Complex aqueous solutions are either irradiated with light (with specific parameters) to form radicals, or light exclusion is performed for the formation of non-radical types of species in the aqueous solution, in order to enable a tailored switching of the adsorption materials. As a further switching parameter, the pH value of the aqueous solution can be increased in order to deprotonate the adsorption materials, or the pH values can be decreased to protonate the adsorption materials, depending on the desired switching events. Light irradiation or light exclusion and/or pH value is set in dependence of the metal cations and/or metal-containing anions, in order to be adsorbed by the adsorption materials. Hence, depending on specific switching parameters, a tailored adsorption and separation of various metal cations and metal-containing anions from complex solutions is achievable with relatively less effort (see examples in Figure 1).
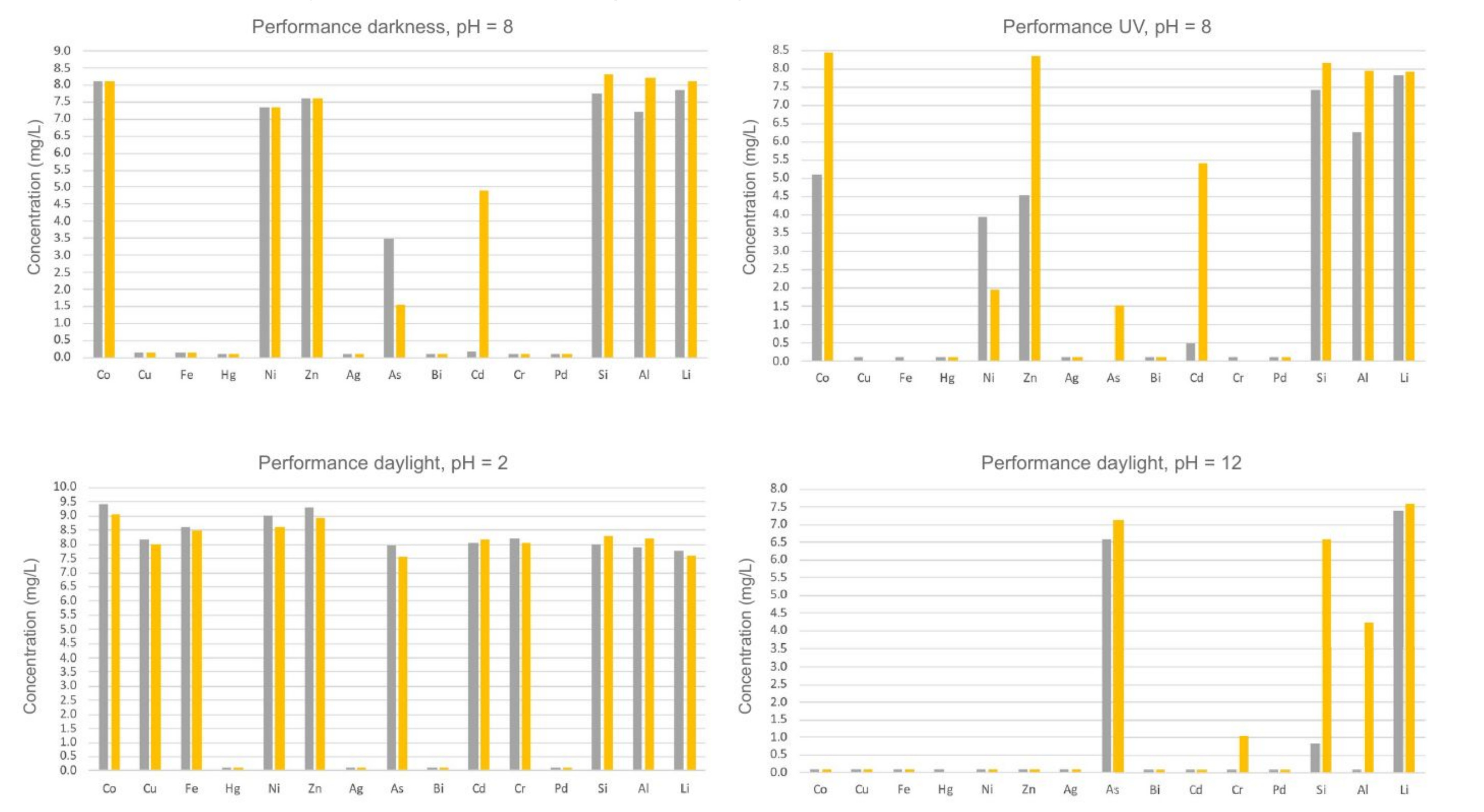 Figure 1: Exemplarily demonstration of selectivities of different adsorption materials in the complexation from metal cation mixtures under variation of the pH value and light conditions, according to the present invention and patent applications (source: Prof. Schmidt and co-workers, Clausthal University of Technology).
Figure 1: Exemplarily demonstration of selectivities of different adsorption materials in the complexation from metal cation mixtures under variation of the pH value and light conditions, according to the present invention and patent applications (source: Prof. Schmidt and co-workers, Clausthal University of Technology).
Advantages
- Adsorption materials are highly stable, regenerative and producable in large quantities at low costs
- Sustainable and economically advantageous technology
- Selective adsorption and efficient separation performance in complex aqueous solutions
- Combination with tailored light sources (e.g. lasers with specific wavelenghts) or ion exchange technologies/li>
Applications
- Selective adsorption and separation of metal cations and metal-containing anions from complex solutions
- Recycling of electronics
- Wastewater and process water treatment
Development Status
The patented technology offers material/compound protection in combination with process protection. The process technology is efficient and reproducible on laboratory scale. Process scale-up is in progress.
Patent Status
European patent application: EP13170042.8
International patent application: PCT/EP2024/058277
Patent holder: Clausthal University of Technology
Contact
Dr. Mirza Mackovic
Patent & Innovation Manager Technology
E-Mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Tel.: +49 551 30724 153
Reference: CPA-2408-T291
Tags: Synthesis and Process Technology, Polymer Chemistry, Chemistry, Physics and Technology & Software
