Novel Channelrhodopsin with increased and stabilized photocurrent
By structure guided rational mutagenesis a superior Channelrhodopsin variant (ChReef) was developed from the Protein ChRmine. A desensitization effect could be reduced by a factor of three and at the same time the light induced photo-current of these variants is five times higher. This allows for an activation of neurons with lower light doses and/or a reduced expression level, which will reduce side effects.
Challenge
Channelrhodopsins (ChRs) enable the control of the activity of excitable cells, like neurons and cardiomyocytes, with light. To do this two steps are needed: (I) the ChRs have to be expressed in the tissue e.g. by transfection with AAV vectors and (ii) a light source has to be implanted. ChRs are widely used in scientific research and have already shown their potential in therapeutic approaches to treat for example retinal degeneration, or for restoration of hearing. But also their use in diseases like Alzheimer or Epilepsy have been shown to be targets for optogenetic therapies. Multiple clinical trials using ChRs are already running (e.g.: NCT04120883, NCT04417348, NCT04945772…). However, the clinical translation of optogenetics remains challenging, since high expression levels and/or the illumination with intense light are needed to activate neurons. This may cause significant side effects and/or result in larger implants, which will make some therapeutic approaches impossible. More efficient ChRs are needed, which allow an induction of high and a stable single channel photocurrent at low light intensities.
Our Solution
Scientists from the University of Göttingen developed the superior Channelrhodopsin-Variant ChReef from the novel Protein ChRmine. ChReef outperforms the state of the art Channelrhodopsin ChrimsonR by a factor of nine in photocurrent output (Fig. 1). ChReef-expression in the optic nerve restored visual function in blind mice with light sources as weak as an iPad screen.

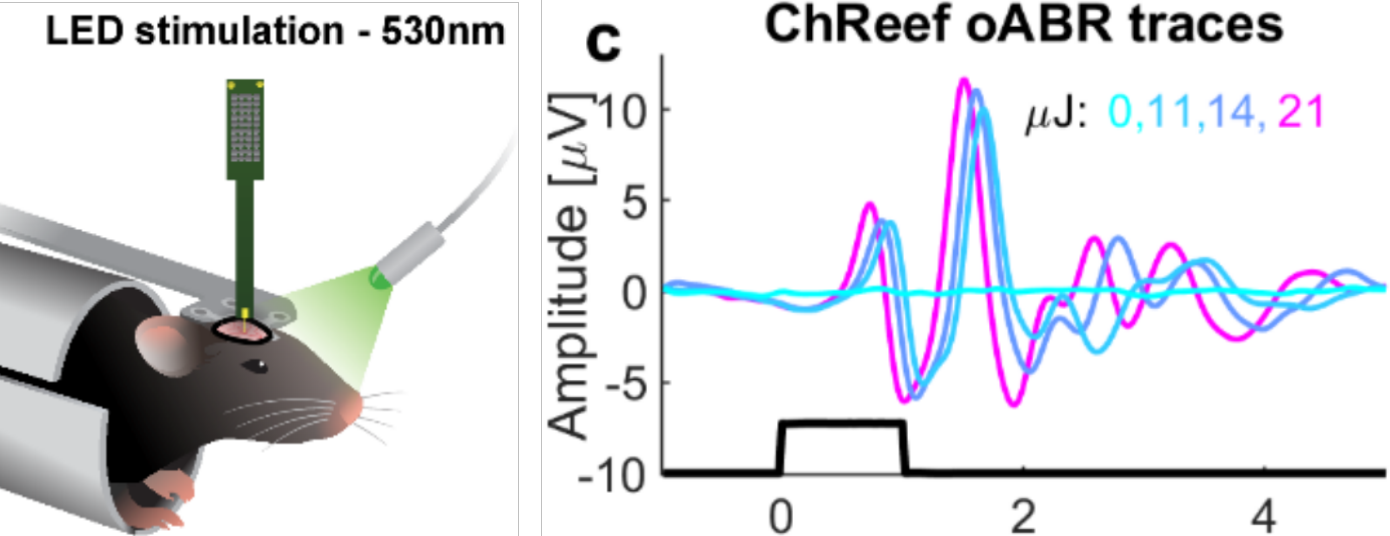
Fig. 1 top) Comparison of ChReef, ChRmine and ChrimsonR: Stationary photocurrent densities. Channelrhodopsins were transiently expressed in NG108-15 cells using the mammalian expression vector pcDNA3.1(-). Cells were illuminated with a 2 s light pulse (λ=532nm) at a saturating light intensity of 23 mW/mm2. Whole cell patch clamp experiments were performed at 60 mV. Fig. 1 bottom) Left:Cartoon depicting photostimulation of mice with a 530 nm LED. Right: oABRs driven with varying radiant flux (1 ms pulses at 10 Hz, colors code the radiant flux in mW) for blind mice injected with AAV2/9-hSynChReef-ES-EYFP-TS
That boost in efficiency enables optogenetic activation with reduced light intensities and/or lower expression levels, which will allow its use for completely new applications and therapeutic approaches.
Advantages
- ChReef outperforms state of the art ChrimsonR by a factor of nine in current output
- ChReef allows restoration of vision in blind mice at roughly one order of magnitude lower light intensities
- Very high single Channel efficiencies allow the use of less light or lower expression-levels
- Less side effects possible in optogenetic gene therapies due to lower expression levels
- High light sensitivity and very high and stable photocurrent allow activation of Neurons inside mammalian tissue
Applications
- Restoration of vision caused by Retinitis Pigmentosa (clincal phases are running)
- Restoration of hearing in mice (preclinical proof of concept in mice)
- Treatment of neurodegenerative diseases like Alzheimer (preclinical proof of concept in mice)
- Drug candidate and drug target identification in cell-cuĺture, organoids and animals
- Research tool
- ChReef may allow the use of optogentics in new therapeutic approaches
Development Status
ChReef has shown to outperform state of the art ChRs in cell culture
Patent Status
Pending patent applications: US and EP
Applicant: University Medical Center of Göttingen, OptoGenTech GmbH
Contact
Dr. Martin Andresen
Patent Manager Life Sciences
E-Mail: Diese E-Mail-Adresse ist vor Spambots geschützt! Zur Anzeige muss JavaScript eingeschaltet sein!
Tel.: +49 551 30724 150
Reference: BioT-2366-UMG
Tags: Opsin, Schwerhörigkeit, Retina Pigmentosa, Gentherapy, kanaldopsin, Optogenetik, Neuron, Rhodopsin, Optogenetic, Life science
